Comprehensive Financial Management for Clinical Trials
finkyn is the one-stop digital solution for the financial management of your trials. While you focus on advancing life-saving therapies, our technology-enabled services will eliminate administrative burdens. Learn more on how finklyn easily integrates into your workflows.
Our Services

Site Payments
Easily integrate contract and eCRF data to automatically pay sites and vendors. Our automated solution streamlines the entire payment process and provides maximum visibility into your trial budgets.

Patient Payments
Provide sites with the ability to easily pay patients right after completed visits to promote retention. Get rid of manual paper-work, outdated cheques and save time on generating transaction reports.

Expertise
Our legal and tax experts support you in setting up your trial contracts in the most efficient way regarding international tax regulations. We also leverage our unique domain know-how to improve your current financial processes.
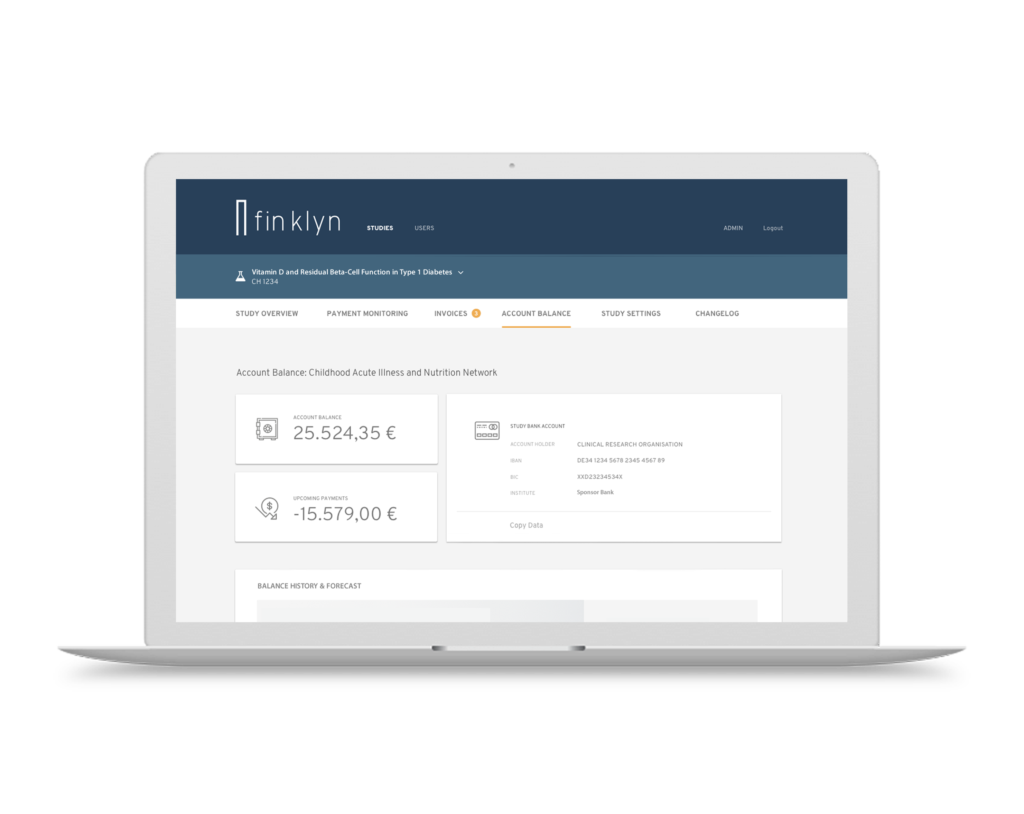
Advanced Technology Platform
Our cloud platform centralizes global payment processes for Sponsor, CROs and Sites. It integrates easily with third-party accounting and eClinical systems systems to create maximum efficiency. With finklyn you can access your information at anytime, anywhere and from any device.
Key Benefits

Efficient Processes
Automated workflows enable you to focus on what really matters in your clinical trial.

Fast implementation
Our digital solution easily integrates with your existing IT and process environment.

Ad-hoc Insights
No more time-consuming reforecasting needed – you instantly know where your study is heading financially.

Data Security
Data storage and processing in Europe ensures maximum data protection according to GDPR.

Simple Interface
Intuitive user interfaces require minimum training effort and keep users happy.

Full Transparency
A comprehensive study dashboard and HCP–compliant reports offer a new level of transparency.
finklyn for you

Sponsor
- Gain instant visibility into financial processes and payment flows.
- Save resources and eliminate errors by automating financial management.
- Increase site satisfaction by simplifying payment workflows.

CRO
- Become a preferred partner for sponsors and sites by offering cutting-edge digital solutions.
- Gain in-depth financial insights and ensure compliant reporting.
- Minimize hours spent on compiling data from multiple systems by using our integrated solution.

Site
- Improve cash liquidity with timely and correct payments after service completion.
- Reduce administrative burdens and manual workflows.
- Gain full visibility on past and upcoming payments for easy cash management.
Selected Partners and Clients







Compliance
finklyn operates according to the risk-based approach of the GAMP-5 guideline. Our way of working is aligned with the following guidelines and standards
- ICH-GCP Guideline
- GMP – Annex 11 “Computerised Systems”
- General Data Protection Regulation (GDPR)
- Internal SOPs, as well as specific SOPs of our customer projects
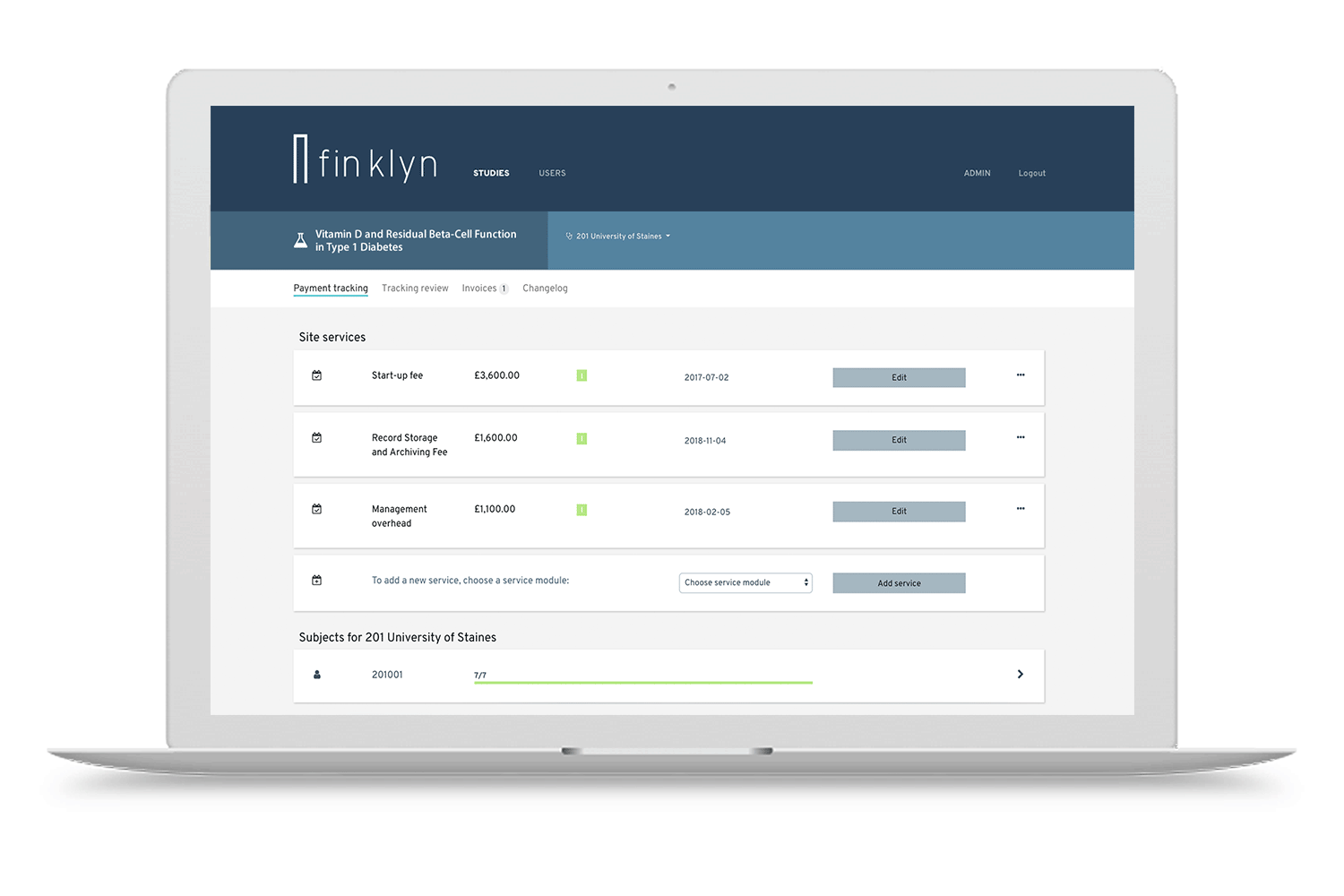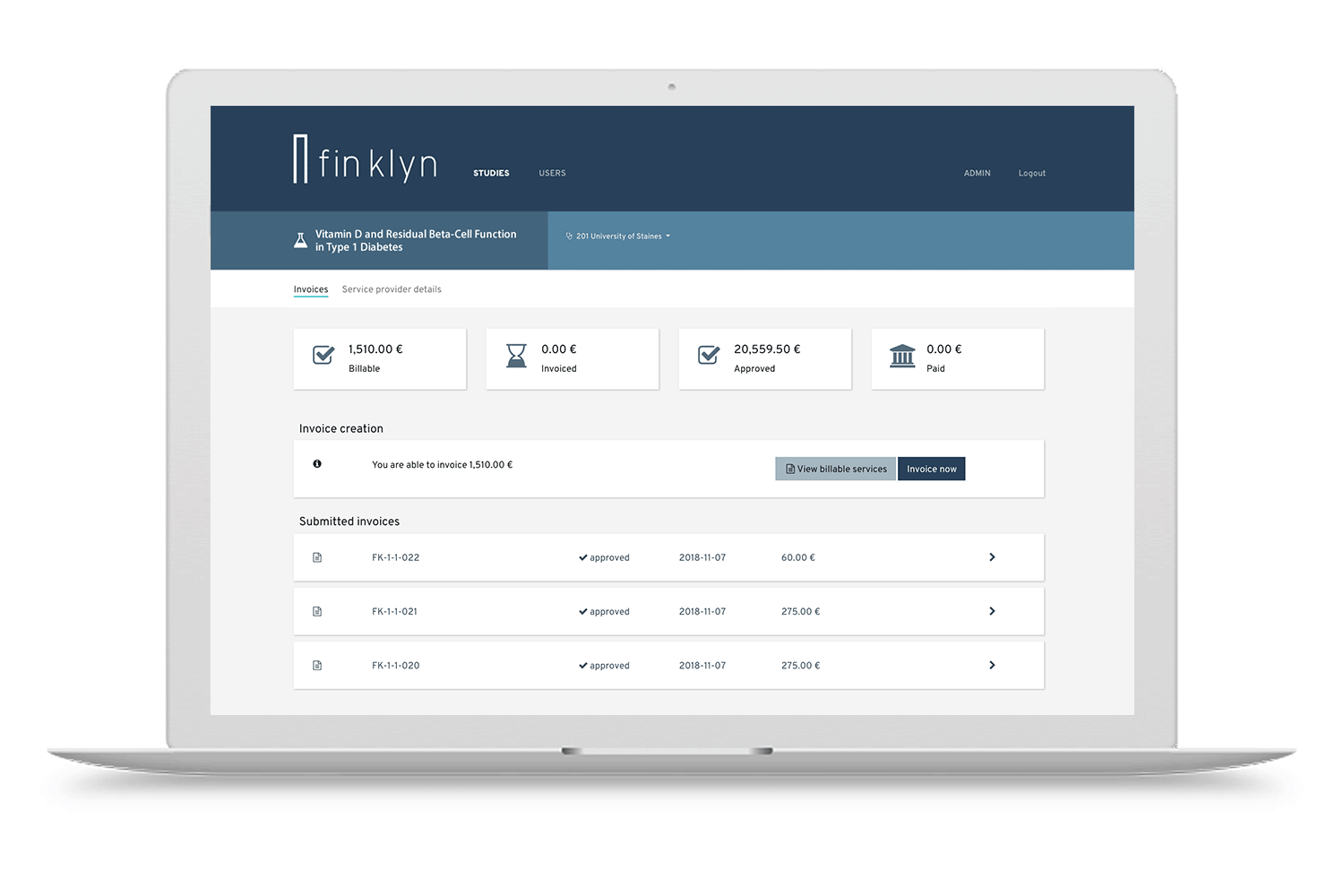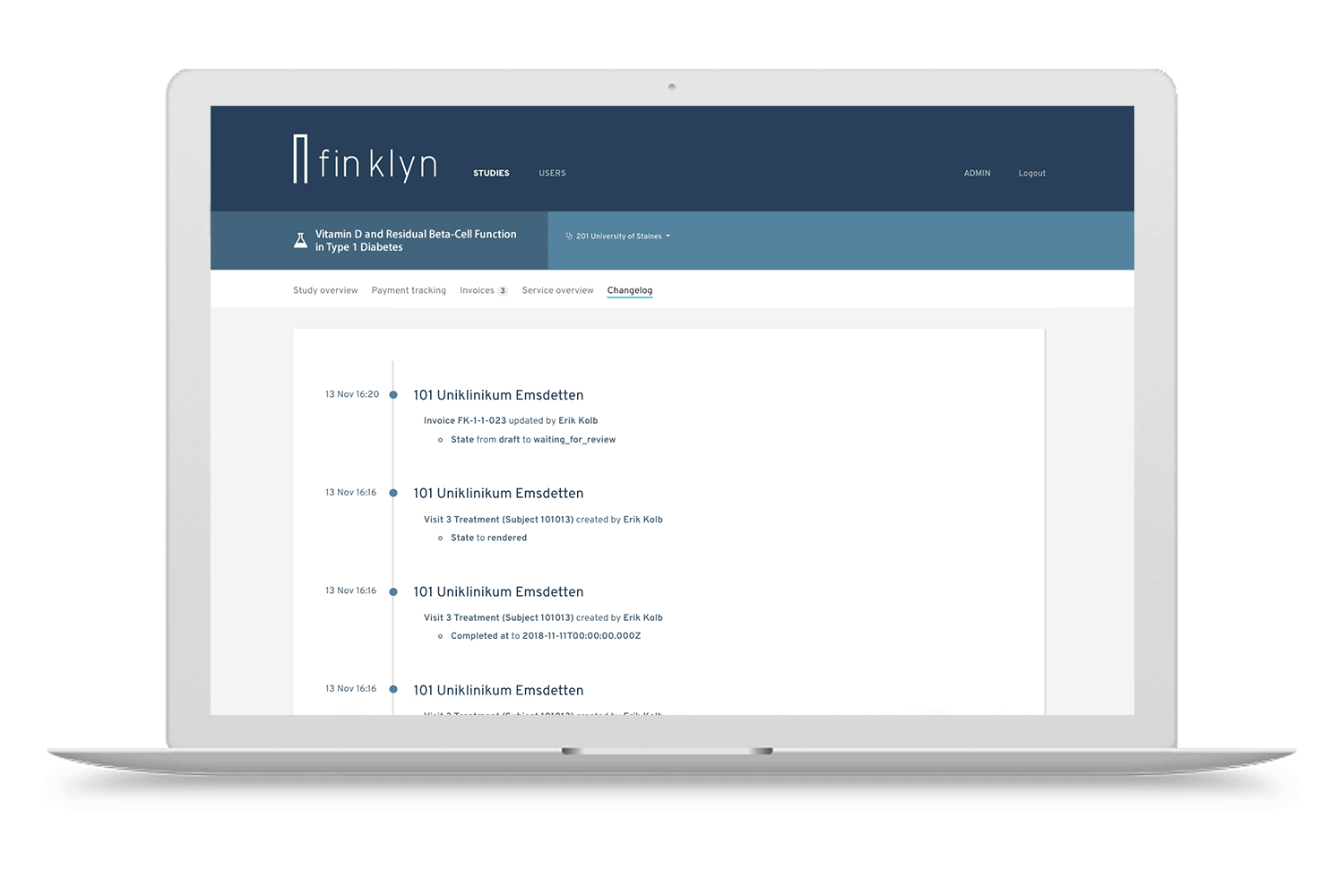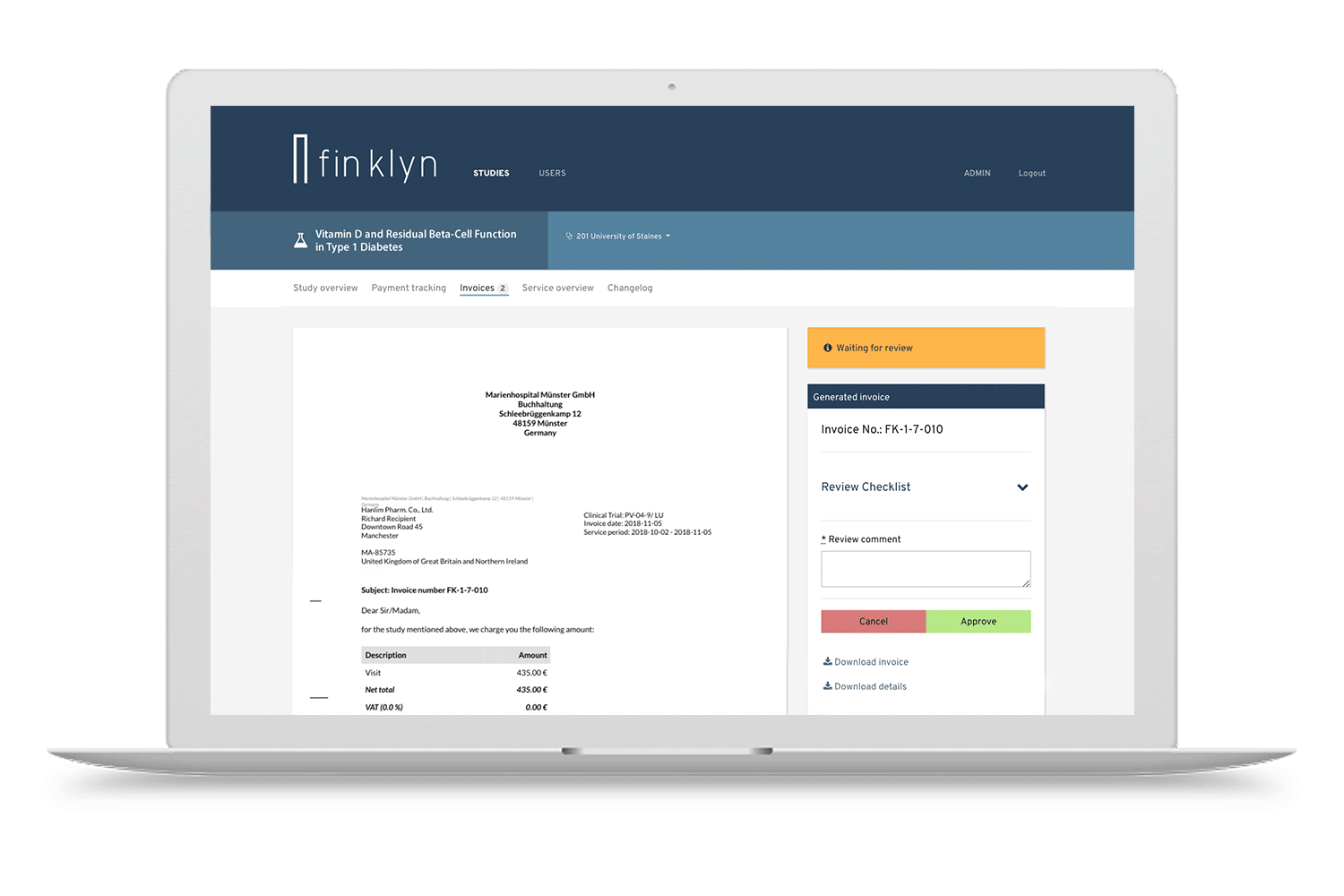
Discover the finklyn platform
Give it a try with a guided tour. Get a first impression on how finklyn can improve your clinical trial operations.
